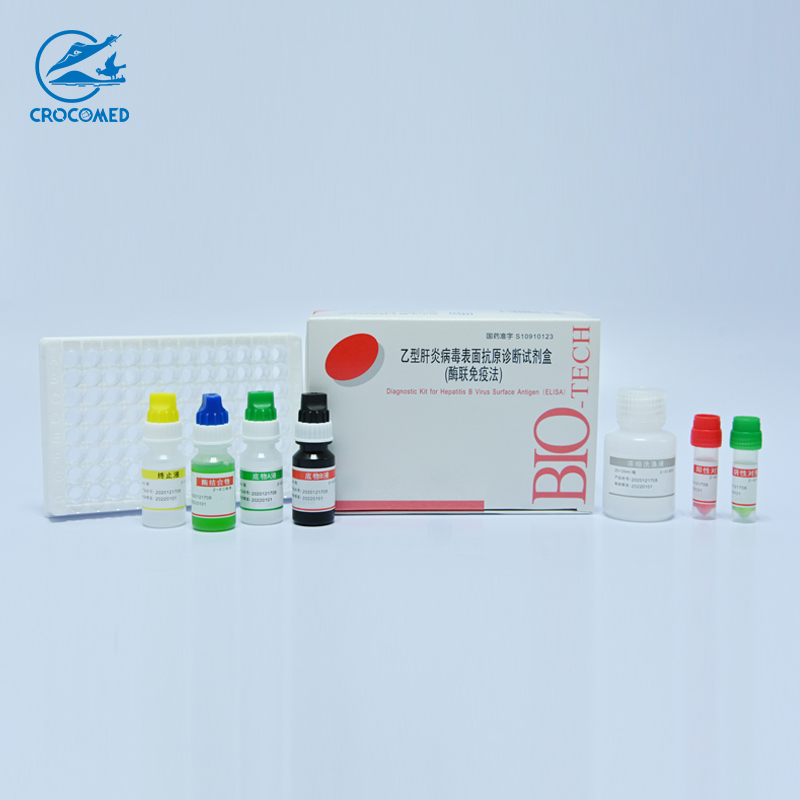
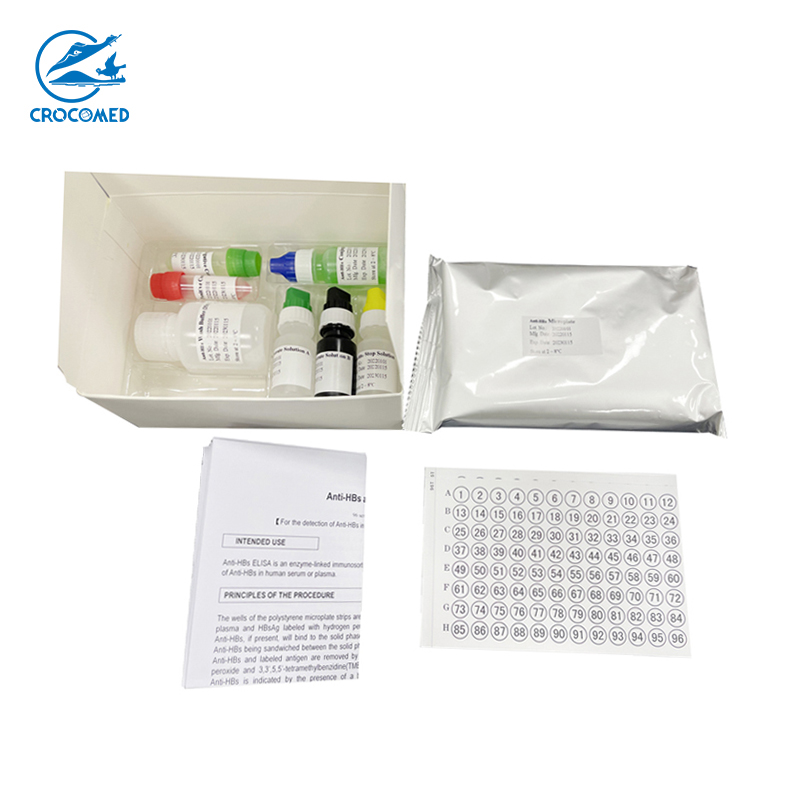
Technical Specifications
1. Microplate 1 plate (96 wells). coated with HBsAg.
2. Positive Control 1 vial (0.5ml). Inactivated Human serum containing Anti-HBs. Preservative: 0.1%ProClin300.
3. Negative Control 1 vial (0.5ml). Inactivated Human serum without Anti-HBs. Preservative: 0.02%NaN 3 .
4. Conjugate 1 vial (6ml). The solution contains HRP-labeled HBsAg, with PBS (0.01mol/L) and stabilizing proteins, ready to use.
5. Wash Buffer (25´ ´) 1 vial (20ml). Diluted 25-fold in distilled water as described in section of Preparation of Reagent.
6. Substrate Solution A 1 vial (6ml). Hydrogen peroxide-urea (approx. 0.6g/L) in citrate-acetate buffer solution (10 mmol/L).
7. Substrate solution B 1 vial (6ml). Tetramethyl benzidine dihydrochloride (10g/L).
8. Stop Solution 1 vial (6ml). 2N sulphuric acid.
9. Plate Covers 2 pieces
10. Instruction Manual 1 copy
Product Advantage
incubated in these coated wells. Anti-HBs, if present, will bind to the solid phase antigen and the labeled antigen resulting in the Anti-HBs being sandwiched
between the solid phase antigen and labeled antigen. Excess unbound Anti-HBs and labeled antigen are removed by washing. Substrate solution containing hydrogen peroxide and 3,3’,5,5’-tetramethylbenzidine(TMB) is then added to each well. The presence of Anti-HBs is indicated by the presence of a blue colour after substrate addition. Reaction is terminated by addition of sulphuric acid. The intensity of the colour is measured spectrophotometrically at 450nm and is proportional to the amount of Anti-HBs present in the specimen.
Product Description
-
Our Human hepatitis B virus surface IgM,HBs-IgM ELISA Kit is to assay HBs-IgM ELISA Kit levels in serum, plasma, culture media or any biological fluid.


FAQ
-
1.We are based in Guangdong, China, start from 2021,sell to Africa(30.00%),South Asia(15.00%),Mid East(10.00%),Domestic Market(10.00%),North America(5.00%),South America(5.00%),Eastern Europe(5.00%),Southeast Asia(5.00%),Oceania(5.00%),Eastern Asia(5.00%),Western Europe(5.00%),Central America(5.00%),Northern Europe(5.00%),Southern Europe(5.00%). There are total about 51-100 staffs in our office.
2. How can we guarantee quality?
Always a pre-production sample before mass production;
Always final Inspection before shipment;
3.What can you buy from us?
Test kit,In Vitro Diganositc,Disposable Samplers,Rapid test kits,Swab
4. Why should you buy from us not from other suppliers?
We focus on producing virus nucleic acid detection kits, Disposable samplers, IGG/IGM rapid test kit, PCR test kit etc.The products approved CE, FDA, SGS, ISO13485.More than 200 staffs, 10 production line.
5. What services can we provide?
Accepted Delivery Terms: FOB,CFR,CIF,EXW;
Accepted Payment Currency:USD,EUR,HKD,CNY;
Accepted Payment Type: T/T,Credit Card,PayPal, Western Union,Escrow;
Language Spoken:English,Chinese,Spanish,Japanese,Portuguese,German,French,Russian,Korean
6.Can I have samples of this goods to test the quality?
Yes, free sample is available.
7.What's the delivery time and can you lower the price?
It depends on your order. The delivery time is within 3-4 days. If you have large quantities,the price and delivery time can be negotiable.
8.Can I customize the packaging and the logo?
Yes, we could offer customized logo for you.
9.What's the shipping method?
We often use DHL, FedEx, UPS, TK,EK,LH to ship our products.
10. The PCR test kit is required to be stored at a temperature between -25 degrees Celsius and -15 degrees Celsius. How did you do it?
Most of our PCR detection reagents are transported in an incubator, and the temperature can be maintained at -20 degrees Celsius for about 12 days, which can fully meet the general air transportation time limit.